Rhizoctonia
Disease Summary Report
Rhizoctonia Solani
General Information
- Broad host range of more than 500 crop and weed species that spans worldwide.
- Survives indefinitely in soil as fine fungal strands.
- Most common in soils of low fertility and microbial growth.
- Trace element deficiencies can increase susceptibility of crops.
- Higher rate of incidence in fields with reduced tillage.
- Barley more susceptible than oats.
- Responsible for stem and stolon canker in potatoes, contributes to pod rot in peanuts.
- Rhyzoctonia, along with Fusarium and Pythium are major contributors to “damping off” seedling disease.
Disease Cycle
- Does not posses ability to produce asexual spores. Produces sexual spores during times of high moisture when environmental conditions are favorable for survival.
- Primarily exists in the soil in the form of mycelia.
- Mycelia feed on dead plants and organic matter (saprophytic) until suitable live tissue from available host roots become available. Mycelia are initially clear or white, and turn tan or brown as growth increases and thickens.
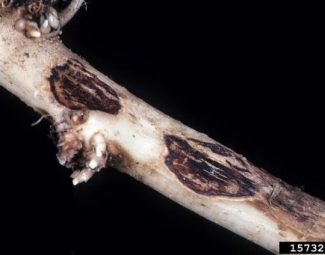
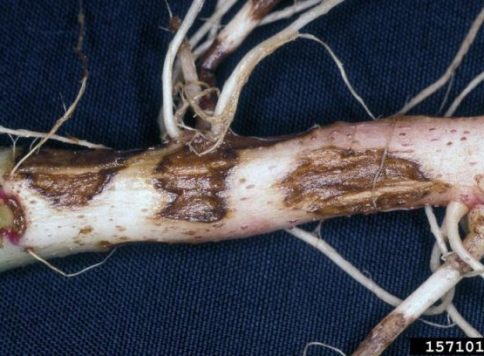
- Infect host by colonizing seeds or young seedling roots. Hyphae from mycelium penetrate hosts and produce enzymes that break down host tissue. Surviving plants may form reddish-brown canker rots on stems and/or roots.
- Produce sexual spores that are dispersed by wind.
- Hyphae that grow from spores fuse with one another to create strands of mycelium.
- Can infect vegetative and fruiting structures that lie on or are in close proximity to soil. Sexual spores can cause rotting of structures higher up on plants during times of high moisture conditions.
Over wintering Strategy
Survive during periods detrimental to growth by producing strands of mycelia called “sclerotia”. Sclerotia are brown to black in color and are capable of surviving many years in near dormancy.
Organic Control
- Commonly Used Control Products
- Myco Seed Treat (Seed Treatment)
- Bac Pac (Seed Treatment)
- NRG Activate PMSLA (Biological)
- NRG Activate 2004 (Biological)
- AgriEnergy SP-1 (Biological)
- Actinovate (Biological)
- Terra Clean (Soil Sterilizer)
- Residuce (Crop Residue Digester)
Cultural Control
- Maintain good fertility and mineral balance in plants. Identify macro- and micro- nutrient deficiencies by performing plant tissue and soil analysis. Adjust for deficiencies with foliar and soil applied applications of appropriate fertilizers.
- Avoid or minimize factors that contribute to proliferation of inoculation such as: inoculated irrigation tail water from infected fields, poor drainage, dirty machinery, wind, erosion, transplants and seeds that are infected with Rhizoctonia, and mechanical and/or insect root injury.
- Create conditions for maximum breakdown of organic residues before planting to reduce additional inoculation and propagation sites.
- Tillage before planting has been shown to reduce rates of infection.
- Plant in areas of good drainage and aeration. Avoid planting densities that inhibit good air movement.
- If possible, remove symptomatic plants as soon as they are identified.
- Plant cultivars that have been identified as being relatively Rhizoctonia resistant.
- Rotate to species of plants that are more Rhizoctonia resistant.
More Images of Rhizoctonia
Bibliography
- Image by University of Georgia Plant Pathology, University of Georgia, Bugwood.org (symptoms on tomato plant)
- Images by Gerald Holmes, California Polytechnic State University at San Luis Obispo, Bugwood.org (bottom rot of lettuce, belly rot of cucumber, lesions on potato roots, watermelon transplant, and scabby lesions on the ground scar of a watermelon, red potatoes, and lesions on lower stem of pepper plant)
- Image by Oliver T. Neher, The Almalgamated Sugar Company, Bugwood.org (root symptoms of rhizoctonia crown and root rot)
- Image by Michelle Grobowski, University of Minnesota Extension – Horticulture, Bugwood.org (pythium spp. and rhizoctonia solani symptoms on cucumber)
- Image by Howard F. Schwartz, Colorado State University, Bugwood.org (early death of potato caused by rhizoctonia solani in the field)
- Farr, D. F., G. F. Bills, G. P. Chamuris, and A. Y. Rossman. 1989. Fungi on Plants and Plant Products in the United States. APS Press. St. Paul, Minnesota. 1252 pp. (Host species)